Mastitis & Congested udder
What we did to identify mastitis & the procedure.
(How to milk sterile & get a sterile milk testing sample)
1) When we suspect mastitis, we do CMT milk testing at the milking.
If we see liquid texture changes into jelly condition, most likely she has mastitis.
2) Checking the temperature and watch how much she is in pain.
We could give banamine (Flunixin) but make sure to give enough liquid so kidney will not get damaged.
How to milk sterile & get a sterile milk testing sample
3) Clean the udder well with alcohol really well especially orifice area.
Several quirts of milk to clear the plug and orifice then milk into the syringe with needle without touching anywhere.
4) Push the needle into the blood sample vial to let the collected milk to go in.
Giving first set of antibiotics while waiting for the milk culture result
5) Milk out completely, finish all the milking routine with her teats cleaning and dip.
6) Take off the cows cap of the ToDay and put smaller tip into her orifice.
Press from the top of the teat and squeeze all into her teat.
Massage her teat to spread the ToDay inside.
7) If the udder is cracked etc., we can put some ointment on the teat. First, we make sure that it is very clean. We put T shirt on so that area is not get covered with dirt etc.
8) Milk her often to flash her bacteria out. (3, 4 ,5 times a day?) Separate babies completely.
Milk her alone and at the last from everyone in order to avoid contaminations.
9) At the last milking in the evening, we can give ToDay. We most likely keep giving ToDay for 5 consecutive days. ToDay's milk withdraw time is 1 week.
If ToDay managed to control the mastitis while waiting for the result of the milk culture test, we can finish the corse of antibiotics.
(How to milk sterile & get a sterile milk testing sample)
1) When we suspect mastitis, we do CMT milk testing at the milking.
If we see liquid texture changes into jelly condition, most likely she has mastitis.
2) Checking the temperature and watch how much she is in pain.
We could give banamine (Flunixin) but make sure to give enough liquid so kidney will not get damaged.
How to milk sterile & get a sterile milk testing sample
3) Clean the udder well with alcohol really well especially orifice area.
Several quirts of milk to clear the plug and orifice then milk into the syringe with needle without touching anywhere.
4) Push the needle into the blood sample vial to let the collected milk to go in.
Giving first set of antibiotics while waiting for the milk culture result
5) Milk out completely, finish all the milking routine with her teats cleaning and dip.
6) Take off the cows cap of the ToDay and put smaller tip into her orifice.
Press from the top of the teat and squeeze all into her teat.
Massage her teat to spread the ToDay inside.
7) If the udder is cracked etc., we can put some ointment on the teat. First, we make sure that it is very clean. We put T shirt on so that area is not get covered with dirt etc.
8) Milk her often to flash her bacteria out. (3, 4 ,5 times a day?) Separate babies completely.
Milk her alone and at the last from everyone in order to avoid contaminations.
9) At the last milking in the evening, we can give ToDay. We most likely keep giving ToDay for 5 consecutive days. ToDay's milk withdraw time is 1 week.
If ToDay managed to control the mastitis while waiting for the result of the milk culture test, we can finish the corse of antibiotics.
About Mastitis
By Michigan State University
Mastitis is an inflammation of udder. Physical injury, stress, or bacteria can cause mastitis. There are several bacteria which are known to cause mastitis in sheep and goats including Streptococcus sp., Staphylococcus sp., Pasteurella sp., and coliforms, such as E. coli. The exact type of bacteria that is causing the mastitis can only be determined by laboratory analysis. Mastitis can either be clinical or subclinical. Clots or serum in the milk are signs of clinical mastitis. In addition the udder may become swollen, hot and/or tender to the touch. Subclinical mastitis is only detectable using a test such as the California Mastitis Test(CMT) or counting inflammatory cells in the milk or culturing milk in the laboratory.
When bacteria enters the udder, a mastitis infection may occur. Good udder and teat conformation can help to reduce the risk of mastitis. One way to help prevent mastitis is to keep milking and living areas clean. Preventing respiratory disease in lambs and nursing kids can also help prevent mastitis and Pasteurella hemolytica, a bacteria that causes pneumonia in lambs and kids can cause mastitis. Also post dipping teats after milking can greatly reduce the risk of mastitis in milking does and ewes. Treatment of mastitis is generally done with the use of either injectable or intramammary antibiotics.
By Michigan State University
Mastitis is an inflammation of udder. Physical injury, stress, or bacteria can cause mastitis. There are several bacteria which are known to cause mastitis in sheep and goats including Streptococcus sp., Staphylococcus sp., Pasteurella sp., and coliforms, such as E. coli. The exact type of bacteria that is causing the mastitis can only be determined by laboratory analysis. Mastitis can either be clinical or subclinical. Clots or serum in the milk are signs of clinical mastitis. In addition the udder may become swollen, hot and/or tender to the touch. Subclinical mastitis is only detectable using a test such as the California Mastitis Test(CMT) or counting inflammatory cells in the milk or culturing milk in the laboratory.
When bacteria enters the udder, a mastitis infection may occur. Good udder and teat conformation can help to reduce the risk of mastitis. One way to help prevent mastitis is to keep milking and living areas clean. Preventing respiratory disease in lambs and nursing kids can also help prevent mastitis and Pasteurella hemolytica, a bacteria that causes pneumonia in lambs and kids can cause mastitis. Also post dipping teats after milking can greatly reduce the risk of mastitis in milking does and ewes. Treatment of mastitis is generally done with the use of either injectable or intramammary antibiotics.
More details about mastitis
Mastitis in Goats Also known as Udder inflammation
Mastitis is the term used to describe inflammation of the mammary glands, which may be caused by a number of different micro-organisms, mostly bacteria, but also viruses (e.g., lentivirus or caprine arthritis encephalitis), fungi, and can also be caused by injury. Although of particular importance in dairy goats, mastitis can develop in any type and breed of doe regardless of its milk yield (Harwood, 2006).
Early signs of mastitis include a drop in milk yield, modified milk texture, color, smell and/or taste, lameness, and / or misshapen udders.
Small ruminant bacterial mastitis is often, but not only chronic and contagious, with infection spreading mainly during milking (Bergonier et al., 2003 ).
The pathogens causing mastitis in goats
Clinical Mastitis
Several pathogens can infect the goat udder, but the most severe is mastitis caused by S. aureus. Although sporadic, clinical mastitis caused by S. aureus may result in gangrenous mastitis, characterized by necrotic udder tissue which will eventually cause the udder to fall off, and the animal will die. The severity and painfulness of this disease makes S. aureus-mastitis a threat to animal welfare.
S. aureus has also been detected in many sub-clinical cases, and it is not yet clear how these relate to clinical cases.
Sub-clinical Mastitis (SCM)
In goats, coagulase-negative Staphylococci (CNS; which is made up of many different species of bacteria) are responsible for most cases of sub-clinical mastitis. Sub-clinical mastitis is characterized by reduced milk production, increased somatic cells and bacterial presence in the milk, but it lacks the macroscopic changes typical of the clinical stage.
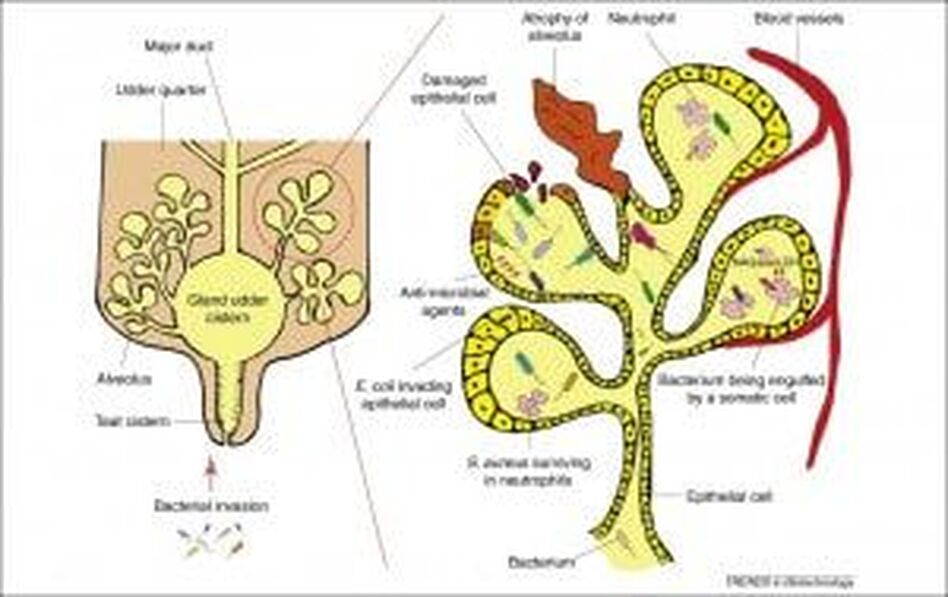
Udder diagram from www.cell.com Trends in Biotechnology
Mastitis is the term used to describe inflammation of the mammary glands, which may be caused by a number of different micro-organisms, mostly bacteria, but also viruses (e.g., lentivirus or caprine arthritis encephalitis), fungi, and can also be caused by injury. Although of particular importance in dairy goats, mastitis can develop in any type and breed of doe regardless of its milk yield (Harwood, 2006).
Early signs of mastitis include a drop in milk yield, modified milk texture, color, smell and/or taste, lameness, and / or misshapen udders.
Small ruminant bacterial mastitis is often, but not only chronic and contagious, with infection spreading mainly during milking (Bergonier et al., 2003 ).
The pathogens causing mastitis in goats
Clinical Mastitis
Several pathogens can infect the goat udder, but the most severe is mastitis caused by S. aureus. Although sporadic, clinical mastitis caused by S. aureus may result in gangrenous mastitis, characterized by necrotic udder tissue which will eventually cause the udder to fall off, and the animal will die. The severity and painfulness of this disease makes S. aureus-mastitis a threat to animal welfare.
S. aureus has also been detected in many sub-clinical cases, and it is not yet clear how these relate to clinical cases.
Sub-clinical Mastitis (SCM)
In goats, coagulase-negative Staphylococci (CNS; which is made up of many different species of bacteria) are responsible for most cases of sub-clinical mastitis. Sub-clinical mastitis is characterized by reduced milk production, increased somatic cells and bacterial presence in the milk, but it lacks the macroscopic changes typical of the clinical stage.
Udder diagram from www.cell.com Trends in Biotechnology
Mastitis occurs when bacteria invades the udder. Good milking hygiene, and good maintenance of the machinery are important in minimizing the amount of bacteria in the parlor and reduce the transmission of pathogens from infected to uninfected udders.
Teat damage
Mastitis develops when bacteria gain access to the udder, via the teat canal (see image above). The teat end can be damaged as a result of over milking, a poorly maintained milking machinery, rough removal of the clusters, getting teats caught on brambles or wire or as a result of teat biting. Teat-end damage allows bacteria to enter the teat canal, and can lead to mastitis.
Viral mastitis
In goats there is a virus that particularly targets the udder, a lentivirus or caprine arthritis encephalitis virus, CAEV. Clinical cases are known as ‘hard udder’. The acute form of this viral mastitis appears at parturition as a very firm udder, but the overlying skin is loose and free from swelling, and most importantly milk flow is almost absent (Koop et al., 2012). Dairy goats that are seropositive for CAEV, and have a negative bacterial culture can still have an increased Somatic Cell Count (See below) (Sánchez et al., 2001). Learn more about CEAV here ).
Somatic Cell CountSomatic cell counts (SCC) is a count of the number of body cells in a quantity of milk expressed as cells/mL, and can be measured at either the gland level (the udder), individual animal level, or herd level (bulk milk SCC) (Koop et al., 2012).
SCCs consist mainly of immune cells (leukocytes) that enter the milk compartment of the udder. There are always small quantities of immune cells in the milk, and their function is to protect the udder against infection by bacteria. Usually, the older the animal gets, the more somatic cells it tends to have in its milk. In dairy cows a relationship between SCC and mammary infections has long been demonstrated, and therefore, SCC are widely used to identify cows likely to have an intra-mammary infection. Most milk processing companies with use bulk milk somatic cell count (BMSCC) to estimate herd mastitis prevalence (Madouasse et al., 2010). However this relationship is a lot more complex in goats.
In goats the relationship between SCC and number of presumed persistently infected does is much harder to define because SCC seems to be affected by infectious and non-infectious factors. (Koop et al., 2012). Several studies have shown that non-infectious factors also influence SCC in goats such as stage of lactation, e.g., the higher the SCC the later the stage of lactation (Gomes et al., 2006; Koop et al., 2011; Olechnowicz and Sobek, 2008), parity (Luengo et al., 2004; Wilson et al., 1995) estrus (Moroni et al., 2007) and breed (Paape et al., 2007).
A single SCC test in goat milk has little value unless stage of lactation and parity is incorporated. So rather than focusing on SCC, it is more effective to keep goats in a clean environment as this will minimize bactoscans. Additionally, hygienic milking, well maintained milking equipment and good milking practice will reduce the incidence of mastitis.
A high SCC in goats is not always accompanied with a positive bacterial culture (Koop et al., 2012). Simply put, the SCC threshold in goats is much higher. Because of this the Californian Mastitis Test (CMT; a crude test for cell counts in milk) should be used with caution in goats. Scientists in the USA recorded SSC from nearly 27,000 goats and found that SCC in goats (and cows) increased with stage of lactation, and parity. By the fifth parity counts for goats were in excess of 1,150,000 cells/ml, exceeding the legal limit of 1,000,000 cells/ml in the US, whereas maximum counts for cows averaged on 300,000 cells/ml (Paape et al., 2007).
Bacteriological culture to test for cause of mastitisBacteriological culture can be used to determine the true infection status of the udder. It can give an indication of which pathogens are present in the herd. This knowledge can be used to guide interventions (Koop et al., 2012).
Diagnosing mastitis in your doesDiagnosis is based on bacterial cultures of milk, and an SCC. However SCC should be interpreted carefully as non-infectious factors can influence the results (See above).
Mastitis develops when bacteria invades the udder. Regular inspection and keeping the teats clean is important in preventing mastitis.
Control and Prevention Nannies affected by mastitis should be isolated, milked last and ultimately culled as this reduces exposure of other does, and increases selection pressure for genetic resistance. In herds where there is a high incidence of mastitis udders should be checked regularly and any lesions present should be treated immediately.
Prevention measures include:
- Improved sanitation – supply plenty of clean and dry bedding
- Hygienic milking practice
- Implementing a milking order, e.g., milk primiparous and / or healthy females first – this has been shown to work in France (Bergonier et al., 2003)
- Dry Period treatment
- Isolating cases
- Culling persistent infectors
Treating MastitisTreatment depends on the severity of the infection. Microorganisms associated with mastitis in dairy goats are commonly controlled with antibiotics, but it is known that continued use of these chemical agents promotes antibiotic resistance among bacterial populations (Gutiérrez-Chávez et al., 2016).
Mild cases may respond to a localized treatment using an intra-mammary preparation of antibiotic into the infected udder, after the teat has been striped out. More severe cases will undoubtedly require more aggressive treatment and you should consult your farm vet (Harwood, 2006).
In New Zealand, researchers looked at the benefits of using CMT as a screening test and concluded that its use resulted in a higher likelihood of finding a gland that would be infected than selecting a gland at random. They also tested treatment (those with a CMT score >1) vs no treatment (Those with a CMT score <1) and found that treatment increased bacteriological cure rate and reduced SCC at gland level compared with no treatment. However, at goat level, milk yield, SCC, and survival were not altered, resulting in no economic benefit of treatment (McDougall et al., 2010).
Good Practice Based on Current KnowledgePreventing mastitis in your herdArdith Mae Farm (AWA Farm) Saanan Does at pasture.
Good milking hygiene will minimize the risk of infection and spread of disease:
- Keep the milking machine serviced
- Use meticulous milking hygiene
- Good husbandry and regular inspection of does udders is essential to avoid suffering so ensure you check udders before milking looking for signs of swelling
- If the goats teats are clean prior to milking avoid washing them, however if they are dirty wash and dry them before milking
- Ensure you pre- or post-dip (which ever works for you) all of the teats
- Treat cases promptly when recognized
- Separate infected and high SCC does into their own milking group and milk last
- Taking samples from all new cases of mastitis is good practice and can help to identify the pattern of infection in a herd and therefore can help in targeting the most effective control measures
- Isolate infected does to avoid spreading the disease throughout the herd
- Cull animals with chronic mastitis, or incurable cases as they will only act as a reservoir for infection
Provision of plenty of clean, dry bedding is required in order to minimise the risk of mastitis. Farmers should assess the cleanliness of their animals regularly and if they are dirty, find out why.
Keep the housing conditions as clean and dry as possible:
- Do not use wet bedding
- Re-bed as often as you can
- Make sure the housing is not overcrowded
- Respond to changes in weather by increasing bedding when very wet
- Avoid water troughs in the bedding area
- Assess the cleanliness of your does regularly
Find the cause of mastitis on your farm
- It is important to know what is causing mastitis in a herd, in order to try and prevent it. The best starting point is to carry out bacteriological examinations.
- Also monitor somatic cell count but remember to take into account the non-infectious factors that can increase SCC in goats (estrus, parity, stage of lactation, stress)
- Decision-making about drying off and culling should be based on SCC recordings and bacteriological examinations
- Name/number of the doe
- Affected teat
- Dates, duration of infection
- Frequency and type of treatment
- Length of the statutory withdrawal period
- Outcome of the treatment (e.g. success/failure/lost quarter/cull/removal to suckling etc.)
Mastitis in Goats References
Gomes, V., Melville Paiva Della Libera, A. M., Paiva, M., Madureira, K. M., & Araújo, W. P. (2006). Effect of the stage of lactation on somatic cell counts in healthy goats (Caprae hircus) breed in Brazil. Small Ruminant Research, 64(1–2), 30–34. https://doi.org/10.1016/j.smallrumres.2005.03.014
Gutiérrez-Chávez, A. J., Martínez-Ortega, E. A., Valencia-Posadas, M., León-Galván, M. F., de la Fuente-Salcido, N. M., Bideshi, D. K., & Barboza-Corona, J. E. (2016). Potential use of Bacillus thuringiensis bacteriocins to control antibiotic-resistant bacteria associated with mastitis in dairy goats. Folia Microbiologica, 61(1), 11–19. https://doi.org/10.1007/s12223-015-0404-0
Harwood, D. (2006). Johne’s Disease (Paratuberculosis / ’MAP’). In Goat health and welfare: a veterinary guide (pp. 96–97). Ramsbury: The Crowood Press. Retrieved from https://www.amazon.co.uk/Goat-Health-Welfare-Veterinary-Guide/dp/1861268246
Harwood, D. (2006). Reproduction and Reproductive Problems. In Goat health and welfare: a veterinary guide (pp. 57–59). Ramsbury: The Crowood Press. Retrieved from https://www.amazon.co.uk/Goat-Health-Welfare-Veterinary-Guide/dp/1861268246
Harwood, D. (2006). The Udder. In Goat health and welfare: a veterinary guide (pp. 143–147). Ramsbury: The Crowood Press. Retrieved from https://www.amazon.co.uk/Goat-Health-Welfare-Veterinary-Guide/dp/1861268246
Karzis, J., Donkin, E. F., & Petzer, I. M. (2007). The influence of intramammary antibiotic treatment, presence of bacteria, stage of lactation and parity in dairy goats as measured by the California Milk Cell Test and somatic cell counts. The Onderstepoort Journal of Veterinary Research, 74(2), 161–167.
Koop, G., van Werven, T., Toft, N., & Nielen, M. (2011). Estimating test characteristics of somatic cell count to detect Staphylococcus aureus-infected dairy goats using latent class analysis. Journal of Dairy Science, 94(6), 2902–2911. https://doi.org/10.3168/jds.2010-3929
Koop, G., Nielen, M., & van Werven, T. (2012). Diagnostic tools to monitor udder health in dairy goats. The Veterinary Quarterly, 32(1), 37–44. https://doi.org/10.1080/01652176.2012.675634
Luengo, C., Sánchez, A., Corrales, J. C., Fernández, C., & Contreras, A. (2004). Influence of intramammary infection and non-infection factors on somatic cell counts in dairy goats. Journal of Dairy Research, 71(2), 169–174. https://doi.org/10.1017/S0022029904000019
McDougall, S., Supré, K., De Vliegher, S., Haesebrouck, F., Hussein, H., Clausen, L., & Prosser, C. (2010). Diagnosis and treatment of subclinical mastitis in early lactation in dairy goats. Journal of Dairy Science, 93(10), 4710–4721. https://doi.org/10.3168/jds.2010-3324
Moroni, P., Pisoni, G., Savoini, G., van Lier, E., Acuña, S., Damián, J. P., & Meikle, A. (2007). Influence of Estrus of Dairy Goats on Somatic Cell Count, Milk Traits, and Sex Steroid Receptors in the Mammary Gland. Journal of Dairy Science, 90(2), 790–797. https://doi.org/10.3168/jds.S0022-0302(07)71563-1
Olechnowicz, J., & Sobek, Z. (2008). Factors of variation influencing production level, SCC and basic milk composition in dairy goats. Journal of Animal and Feed Sciences, 17(1), 41–49. https://doi.org/10.22358/jafs/66468/2008
Paape, M. J., Wiggans, G. R., Bannerman, D. D., Thomas, D. L., Sanders, A. H., Contreras, A., … Miller, R. H. (2007). Monitoring goat and sheep milk somatic cell counts. Small Ruminant Research, 68(1–2), 114–125. https://doi.org/10.1016/j.smallrumres.2006.09.014
Petzer, I. M., Donkin, E. F., Du Preez, E., Karzis, J., van der Schans, T. J., Watermeyer, J. C., & van Reenen, R. (2008). Value of tests for evaluating udder health in dairy goats: somatic cell counts, California Milk Cell Test and electrical conductivity. The Onderstepoort Journal of Veterinary Research, 75(4), 279–287.
Sanchez, A., Contreras, A., Corrales, J. C., & Marco, J. C. (2001). Relationships between infection with caprine arthritis encephalitis virus, intramammary bacterial infection and somatic cell counts in dairy goats. Veterinary Record, 148(23), 711–714. https://doi.org/10.1136/vr.148.23.711
Wilson, D. J., Stewart, K. N., & Sears, P. M. (1995). Effects of stage of lactation, production, parity and season on somatic cell counts in infected and uninfected dairy goats. Small Ruminant Research, 16(2), 165–169. https://doi.org/10.1016/0921-4488(95)00622-R